NEW YORK — Bradley Little, a physical education teacher in Arizona, was leading his class through a school hallway in 2017 when he collapsed. Little feared he was having a stroke. Or, in a sign of the times, that he'd been shot. He tried to stand, but his leg wouldn't move.
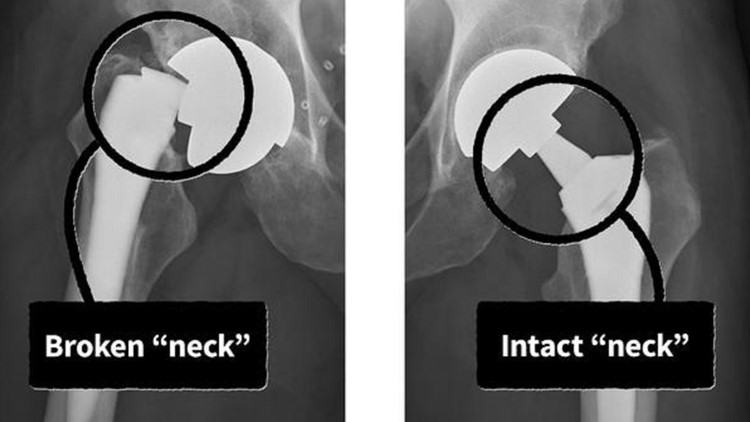
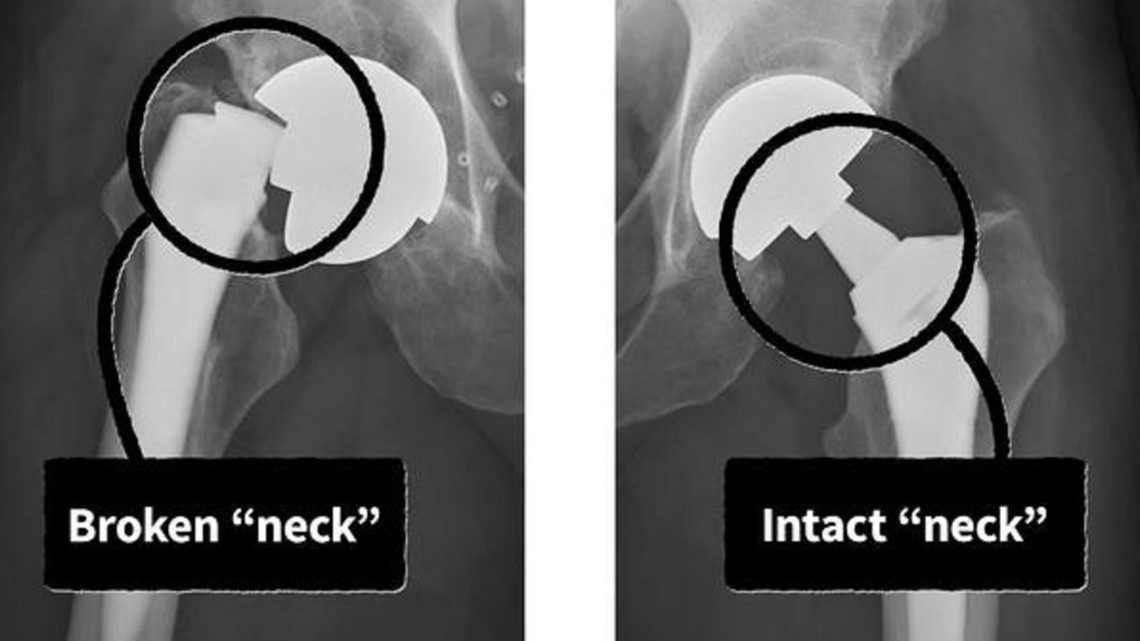
A student ran for help. Firefighters arrived and hoisted Little onto a gurney. At the hospital, an X-ray revealed that the artificial hip implant in Little's right leg had "suddenly and catastrophically structurally failed," according to a lawsuit Little would later file in federal court. The implant severed at its "neck" — a 2-inch-long titanium part linking Little's thigh to his torso.
"It looked like a laser went through it," Little said in an interview. "It was like someone just went in there and cut it right in the middle."


Profemur artificial hips were once considered innovative for a feature known as "dual modular necks," intended to modernize total hip replacement surgery. Hundreds of thousands of Americans undergo hip implant surgery each year and devices are expected to last at least 20 years, according to the American College of Rheumatology. The Profemur necks, available in an array of lengths and angles, made it easier to customize the hip implants for patients.
But the neck also proved to be a weak point: Over the past two decades, more than 750 Profemur hips like Little's have fractured at the neck, an attorney for the manufacturer has said in court. In interviews, patients said they were left unable to walk and in need of emergency surgery. Reports submitted to the FDA describe Profemur patients stranded in the midst of routine life, while hiking, golfing, bowling, mowing the lawn, lifting a potted plant, getting out of a chair, putting on pants, and leaning over to pick up a key.
After each break, patients endure an hours-long repair surgery that can be traumatic because the broken implant is embedded in their bone and difficult to remove, according to three orthopedic surgeons who've performed such a procedure. The repair surgery, which can cost tens of thousands of dollars and may not be fully reimbursed by insurance, often requires a patient's femur to be cracked open to extract a metal stem that was inserted down its length. Lawsuits have likened removing the bone around the stem to peeling a banana.
"It's gruesome," said Lee Eric Rubin, an orthopedic surgeon and expert on prosthetic hips at Yale University. "There's no way around the fact that there's a failed or broken implant in that patient's thigh. We have to remove it."

Many Profemur fractures in patients' bodies could have been avoided if the manufacturer or the FDA responded to early signs of failure with more urgency, according to a months-long investigation by KFF Health News and CBS News. An FDA database shows reports of Profemur's titanium modular necks breaking inside U.S. patients since at least 2005, but the corresponding parts were not recalled until 15 years later, if at all. Ten sizes of the titanium neck eventually were recalled in 2020 after being identified in more than 650 reports of fractures submitted to the FDA. Six other sizes of titanium necks, identified in about 75 additional fracture reports, have not been permanently recalled.
Aidin Eslam Pour, another Yale orthopedic surgeon who has studied Profemur fractures, said the manufacturer "waited too long."
"This implant should have been pulled out of the market earlier," he said.
Profemur's original manufacturer, Wright Medical Technology, in 2009 switched the metal of the modular neck from titanium to a stronger cobalt-chromium alloy, FDA documents show. Then, after some of those necks also began to break, the company recalled one size but left 11 others on the market despite reports of corrosion causing the implants to fail, FDA documents show.
In total, at least 28 sizes of the Profemur artificial hips with a dual modular neck have allegedly fractured or corroded, but just 11 sizes have been permanently recalled, according to FDA data and records.
Wright Medical, a Tennessee company founded in 1950, has made implantable medical devices since at least the 1970s, according to the company website. Wright sold its hip and knee implant division, including the Profemur, to Chinese company MicroPort for $285 million in 2013, according to the Securities and Exchange Commission. Stryker Corp., one of the nation's largest device companies, paid about $4 billion for the rest of Wright in 2020.
Wright Medical declined to comment in an email from Stryker spokesperson Jon Zimmer. MicroPort did not respond to more than a dozen requests for comment sent to its attorneys, public relations firm, and U.S. offices. MicroPort still advertises Profemur hip implants with dual modular necks on its website, where the devices are listed as "not marketed/registered in United States."

The FDA declined to provide an official for an interview and did not answer written questions about why some Profemur sizes were not permanently recalled. In an email, FDA spokesperson Audra Harrison said medical device manufacturers are largely responsible for deciding which products to recall and when to do so, while the agency "monitors" this process and requests recalls only in "urgent situations." In the case of the Profemur modular necks, all recalls were initiated by MicroPort, and the FDA "took action accordingly," the agency said.
For this investigation, journalists with KFF Health News and CBS News analyzed thousands of reports of Profemur complications submitted over the past two decades to the FDA's nationwide MAUDE database, which catalogs reports of medical device problems and malfunctions. MAUDE is unverified, incomplete, and imperfect — for example, not all device problems are properly submitted to the database, and a single issue may be reported more than once. However, the database still offers the best available perspective on medical device complications in the United States. The FDA has used MAUDE to identify device problems since the early '90s.
KFF Health News and CBS News also reviewed about 180 lawsuits filed in federal court in the past decade alleging Profemur modular necks broke or corroded. Plaintiffs have alleged severe pain, swelling, a "debilitating lack of mobility," and, in at least a few cases, nerve damage and neurological issues from cobalt and chromium ions leaking into their bloodstream.
RELATED: Thousands got Exactech knee or hip replacements. Then, patients say, the parts began to fail.
Most of the lawsuits have been resolved through out-of-court settlements without Wright Medical or MicroPort publicly admitting fault, according to court filings. The remainder of the lawsuits are ongoing.
Wright Medical has denied liability in some lawsuits before settling them and has defended Profemur implants in court in the years before some of the implants were recalled for fracturing.
"A device fracture does not mean it is defective," Wright Medical attorney Tiffany Carpenter said in federal court in 2018, according to a hearing transcript. "Devices fracture all the time."
Collectively, the lawsuits allege that Profemur artificial hips broke or corroded at the neck in about 7½ years, on average. Profemur necks made from titanium broke on average in about 10 years while necks made from the cobalt-chromium alloy broke or corroded in just six years, the lawsuits allege.
Some plaintiffs say they got Profemur implants in both legs — then they both ended up breaking.
Mark Feld, 75, of New Hampshire, who was an avid runner, said he was implanted with Profemur artificial hips in his right and left legs in 2005 and 2008, then the right hip fractured within 10 years, according to a lawsuit he filed. Wright Medical denied liability in court filings and settled out of court for an undisclosed amount.


Feld said that because he surrendered all claims against Wright in the settlement, he could not sue again when his left implant broke in 2020 as he was walking across a bridge near his apartment.
He crawled home to call 911, he said, and was rushed to the hospital.
"I couldn't walk across that bridge for a year," Feld said. "And to this day, I still feel like a ticking time bomb. … Nobody could confirm for me that it can't happen again."
Little, the Arizona teacher, also suffered a second Profemur break, four years after his first, according to his lawsuit, in which Wright Medical denied liability and settled out of court. Little said in an interview that this time he was teaching class on a tennis court when he felt a sensation in his left leg that reminded him of crushing an aluminum can. He said he narrowly avoided tumbling onto his students.
After his two broken hip implants and replacement surgeries, Little said, he had to stop coaching basketball and will retire from teaching at the end of this school year — four years earlier than planned. He still feels unsteady and is afraid to climb a stepladder to change a lightbulb, he said.
"I've been robbed of some things," Little said. "There should be accountability for it."
It is not publicly known how many Profemur hips have failed. According to a federal court transcript, Carpenter, the attorney for Wright Medical, said in court in 2018 that the company was aware of 768 fractures among about 353,000 Profemur necks sold. That's a fracture rate of about 0.2%.
Other sources report a much higher rate. The Profemur devices that were permanently recalled in 2020 had a U.S. fracture rate of 2.2% — 11 times what was described in court — according to FDA documents. Peer-reviewed studies estimate fracture rates as low as 1% and as high as 6% for some Profemur models.
Even the lowest estimates are "unacceptable," said Samo Fokter, an orthopedic surgeon and Profemur expert at University Medical Center Maribor in Slovenia.
Fokter has co-authored more than 10 peer-reviewed studies on the Profemur, including one this year, and said he implanted about 50 of them before they were known to fracture.
"This should not happen," Fokter said. "If you put too much force on any implant, it can fracture, of course, but this is very, very rare. Not approaching 1%. It should be less than one in 100,000, let's say."
"Like a black hole developed under their foot"
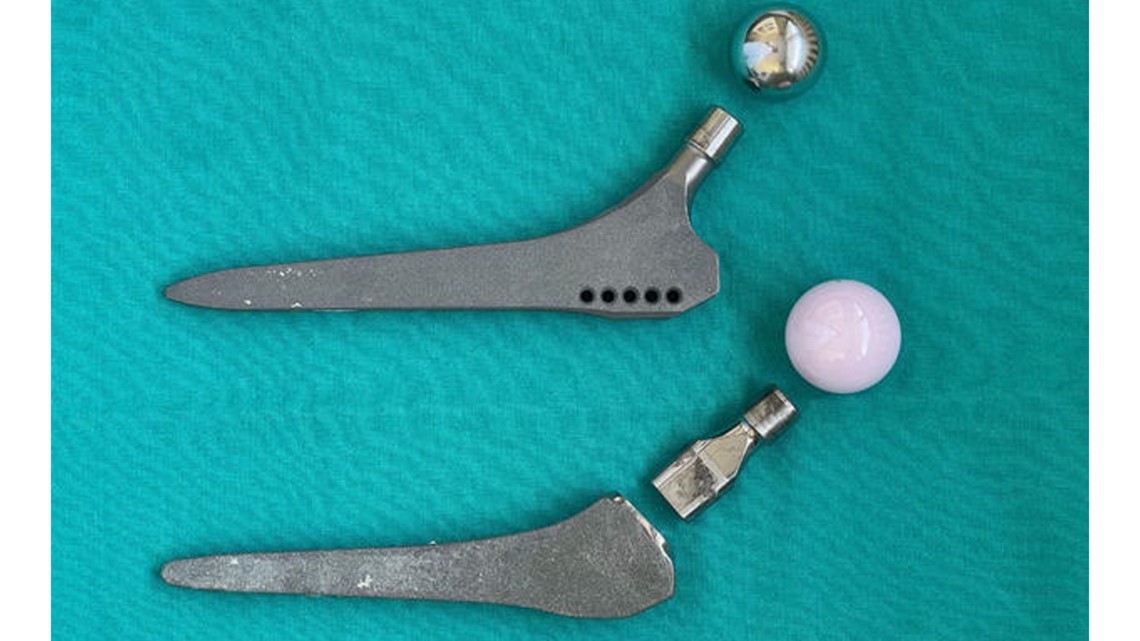
The Profemur's problems originate from its "neck," which is a metal connector between the upper components in the hip socket to a lower "stem" that is inserted into a patient's thigh bone, according to peer-reviewed studies, court records, and expert interviews.
Historically, an artificial hip's stem and neck were a single piece of metal. The Profemur line added a junction at the top of the stem so the neck was separate. Because these dual modular necks detached on both ends, the size and angle could be changed to better fit a patient.
But the Profemur's additional junction was also its downfall. Rubin, one of the Yale experts, who also maintains an exhibit of the history of prosthetic hips at the university, said in some patients tiny cracks formed on the portion of the neck that slotted into the socket of the stem. Patients had no idea their implant was cracking until the neck snapped, he said.
"From a patient's perspective, they're walking around on what otherwise would seem like a successful hip implant," Rubin said. "And all of a sudden, as they took a step, they could not bear weight … like a black hole had developed under their foot."
The dual modular neck was developed by a European company, Cremascoli Ortho Group, in the '80s, then purchased by Wright Medical in 1999 to be introduced as the Profemur in the United States. The Profemur was cleared for sale by the FDA in 2000 through the 510(k) program, which permits new medical devices to be sold without extensive testing if they are deemed to have "substantial equivalence" to other devices already on the market. Through this process, new medical devices can piggyback on a single approval for decades.
Wright Medical told the FDA that the Profemur was substantially equivalent to five existing artificial hip systems, and the agency agreed, according to FDA documents obtained through a Freedom of Information Act request. However, of those five hip systems, at least three had significantly different necks than the Profemur, Rubin said. And one was later recalled because of its high failure rate, according to the FDA.
The FDA documents state that although the Profemur is different from the older hip implants that its approval was based on, those differences were "not expected to affect the device's safety and effectiveness." Spokesperson Harrison said in an email that the FDA "followed the statutory framework" when the Profemur was reviewed and cleared.
Once it was cleared by the FDA, Wright touted the Profemur's dual modular neck as a feature.
In a promotional document obtained by KFF Health News and CBS News, Wright guaranteed the "structural reliability" and "absence of fretting corrosion" at the junction of the stem and neck. Then Wright marketed the Profemur to people with an "active lifestyle," saying the product was for patients who wanted to return to activities like golf, tennis, karate, and wrestling after their hip replacements, according to at least two dozen lawsuits filed against the company.
Wright also hired Jimmy Connors, who was the world's top-ranked tennis player in the '70s, as a spokesperson.
"This hip has given me back my quality of life. It's allowed me to do anything I did before," Connors said on JimmysNEWHip.com, a website launched by Wright in 2006, according to screen captures of the site preserved by the Internet Archive.
When the website launched, Wright Medical knew of at least some reports of modular neck fractures. Multiple lawsuits allege the company was aware as of 2000 that some Cremascoli hips had fractured at the modular neck, and then became aware of more fractures in 2003 and 2004. The FDA database shows Wright was also aware of two Profemur implants that allegedly fractured at the neck and were returned to Wright in spring 2005.
In 2006, FDA data showed six reports of Profemur fractures that identified the neck as the part that allegedly broke. By 2007, there were 11 such reports. By 2008, there were 30.
Connors, reached on his cellphone, said Wright Medical did not inform him of Profemur fractures at the time of his endorsement or since. Connors said his own hip implant did not fracture but had to be replaced in 2012 because of other complications.
If he had been told about a fracture risk, Connors said, he might have chosen another implant.
"If I was going through it now, I'd know a lot more to ask than I did back in the first time," Connors said.
Perry Parks, 79, who played football for the Los Angeles Rams in the '60s, said Connors' endorsement persuaded him to get a Profemur hip in 2007. His implant snapped six years later during a bike ride, according to his lawsuit. Wright Medical denied liability and settled out of court.
In an interview, Parks said he was lucky to be biking at the beach at the time of the break, where he tumbled into sand, instead of in traffic.


"The thing that incenses me more is that they knew this," Parks said. "There was some intentionality here to put … profits over the health of people."
New metal, new complications
In 2009, Wright Medical introduced a new version of the Profemur modular neck that once again was cleared for sale by the FDA. Agency documents show that the neck material was switched from titanium to a cobalt-chromium alloy, a stronger metal.
"That was a big mistake," Fokter said.
While the cobalt-chromium necks were less susceptible to fracture, they created a new problem at the same junction between the neck and stem, said Fokter and the two Yale experts. Once implanted, the cobalt-chromium neck could rub against the stem's titanium socket, leading to a form of bimetallic corrosion that can cause pain and swelling and leak small amounts of metal ions into a patient's bloodstream, potentially causing a long list of complications, the three experts said.
Robert Rembisz, 75, a retiree in Port St. Lucie, Florida, alleged in an ongoing lawsuit that Profemur corrosion in his right leg caused elevated metal levels in his blood and "neurologic symptoms" including nerve damage, tinnitus, and balance and coordination problems. Wright Medical has not yet responded to the allegations in Rembisz's lawsuit.
Rembisz added in an interview that he believes the implant hindered his memory and cognition, leading him to question whether he was suffering early signs of dementia. He provided to KFF Health News and CBS News lab reports showing the metals in his blood rising over years, with cobalt levels peaking at nearly 12 times the normal range. Rembisz said most of the symptoms faded after his implant was removed in 2021.
"The problems I developed weren't even close to my hip," Rembisz said. "This problem could be occurring in [other people's] bodies as well. And they don't even know it."
Six years after Profemur switched metals, MicroPort recalled one size of the cobalt-chromium neck affecting about 10,500 implants, citing an "unexpected rate of postoperative fractures," according to FDA records. But it is unknown how many could not be returned because they'd already been implanted.
Kristin Biorn had one.
Biorn, 74, of Pasadena, California, alleged in a lawsuit that this particular size of Profemur neck was implanted in her left leg in 2013 and broke within two years — four months before the recall. Wright Medical and MicroPort denied liability in her lawsuit, then settled out of court.
In an interview, Biorn said the break occurred as she was working at her burgeoning home-staging business. While putting final touches on a client's home with her teenage son, she fell to the floor, unable to stand or crawl, she said.
"Honestly, it gives me nightmares about what could have happened had my son not been there," Biorn said. "My phone was downstairs and there was no way I could have gotten down the stairs alone. No one was scheduled to come in for four days."


Biorn said in her interview that it took three surgeries to fix her hip after the Profemur fracture and she was ultimately forced to close her business and retire.
She now walks with a cane.
Although MicroPort recalled one cobalt-chromium size in 2015, the company did not recall 11 other sizes made of the same metal with the same design, and some lawsuits have faulted the company for leaving "interchangeable" products on the market. MicroPort also did not at that time recall any of the titanium necks, which as of 2015 were identified in more than 500 fracture reports in FDA's database. MicroPort recalled 10 titanium sizes in 2020.
Finally, also in 2020, MicroPort issued a sweeping recall for all available Profemur modular necks, regardless of whether they were made of titanium or cobalt-chromium, according to FDA records.
The recall was temporary so MicroPort could update the packaging of Profemur implants.
Afterward, the FDA would allow them to go back out for sale.
KFF Health News data editor Holly K. Hacker and CBS News producer Nicole Keller contributed to this report.
KFF Health News, formerly known as Kaiser Health News (KHN), is a national newsroom that produces in-depth journalism about health issues and is one of the core operating programs at KFF — the independent source for health policy research, polling, and journalism.